Who Owns What in SaaS Validation? Vendor vs Regulated Company Responsibilities
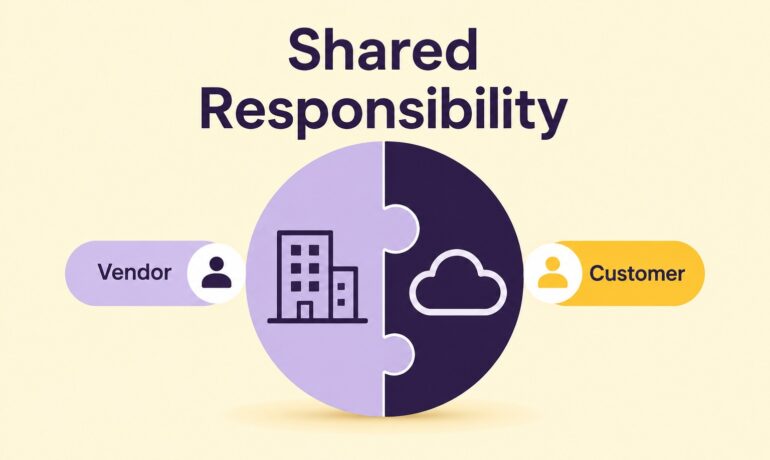
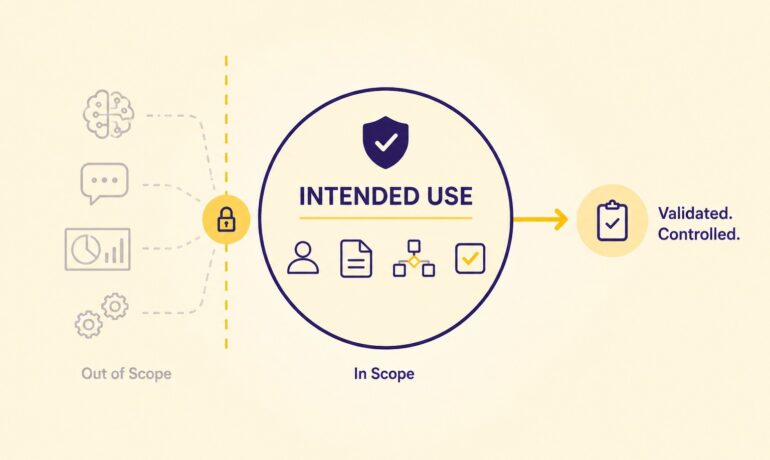
Many life sciences organizations commonly ask this question when implementing a SaaS platform: “If the vendor already validated the system, what do we still need to do?” The answer is straightforward. The vendor is responsible for the platform it develops, operates, and maintains. The regulated company remains responsible for how that platform is configured, governed,